Artificial Intelligence, the transformation of rheumatology? Part II
Estefania Fajardo, Cesar Graf MD
INTRODUCTION
Since the beginning of the XXI century, and most notably since the 2010s, the practice of medicine has been at the center of a great deal of innovation regarding the implementation of digital health technologies. This includes the electronic health record, virtual visits, mobile health, wearable technology, digital therapeutics, artificial intelligence, and machine learning. Currently, the increased availability of these technologies offers opportunities for improving important aspects of the practice of medicine, including access, outcomes, adherence, and research (1).
In this opportunity, we address the application of artificial intelligence (AI) and its different areas in the field of rheumatology, and we also discuss the challenges posed by this incursion into technology that has been developing for years, as we saw in the first part of this series of special articles (2), as well as the tools available and how we can leverage these new forms for the benefit of our patients.
Rheumatologists have substantial experience and a deep contextual understanding of the challenges facing patients, doctors, and healthcare systems. Therefore, they should target the development and applications of this technology to the areas where it will be of greatest benefit.
It is clear that due to the sheer volume of data available in electronic health records, the number of possible treatment and outcome trajectories associated to comorbidities, medications, and other heterogeneous patient factors is greater than what a human being, even an experienced doctor, can analyze.
AI approaches have he potential to transform the treatment of rheumatic diseases, from advancing early diagnosis to facilitating a more individualized treatment approach, with the overall aim of improving patient outcomes. (3,4)
Ultimately, AI will be a key tool on the rheumatologists' team that, among other things, has the potential to support research and clinical care, and perhaps even alleviate some of the bureaucratic burdens. Because of these aspects, it is necessary to be aware of the implications arising from the application of artificial intelligence technologies. (2,3,4).
Just as with the implementation of any new technology, there will be hurdles before the widespread adoption of AI in healthcare. There are currently debates about data privacy, regulations, and technical challenges, so there is a need to review the records and algorithms used to train different AI systems with a critical approach. (5) On this, so far, there is no replacement for human scrutiny and care.
WHERE ARE WE IN RHEUMATOLOGY?
In recent years, we have seen substantial progress in the treatment of rheumatic diseases; we have achieved a better understanding of its physiopathogenesis and the possibility of repositioning the use of antirheumatic medications, designing new therapies, and developing therapeutic strategies such as T2T, initially applied in rheumatoid arthritis, and later in other diseases, such as lupus, spondyloarthritis and other pathologies in which follow-up with tools such as clinimetric have made it possible to optimize the treatment of patients coping with inflammatory arthritis (3,5).
AI has been used in some areas of rheumatology with encouraging results. Some of the areas where great improvements have been achieved include imaging, classification, and disease progression and prediction of response to treatments.
The evolution of artificial intelligence in rheumatology is happening in different phases, perhaps more slowly than for other specialties, but with tangible progress. A first phase has included exploratory projects to validate the approaches to clinical experience, which involves proof-of-concept studies and refinement of existing tools. Further developments are expected in algorithms to improve clinical practice, as these save time in automated image analysis and also advances in algorithms to help guide clinical decisions and, subsequently, algorithms to facilitate a fully personalized treatment approach to improve patient outcomes. (2)
Three subgroups are identified in machine learning (Table 1), and of particular interest are the models developing deep learning (6) (see Figure).
Table 1.
|
Type of learning |
Example |
|
Supervised learning |
Such as handwriting recognition, digit recognition, image and document classification. |
|
Unsupervised learning |
Predictive model trained similarly to supervised learning, but the difference is that learning occurs on unclassified or untagged data, finding patterns of similar examples across datasets. |
|
Reinforcement learning |
Is a type of machine learning method in which there is no training with classified or unclassified data; the system learns in an interactive environment with no information about the possible outcome, by trial and error using feedback from its own actions and experiences. In other words, the model is reinforced by solving the problem in the best possible way (14)
|
Source: Elaborated by the authors
Deep learning can help rheumatologists by classifying diseases and predicting disease activity of each pathology. Automated image recognition and natural language processing will likely pioneer the implementation of artificial intelligence in rheumatology. (4)
There is a long history of artificial intelligence in imaging in rheumatology, using classical AI methods, but most of them did not make it into clinical practice. Recently deep learning tools have shown the potential to automatically interpret images beyond human-level accuracy. (7)
Figure 1
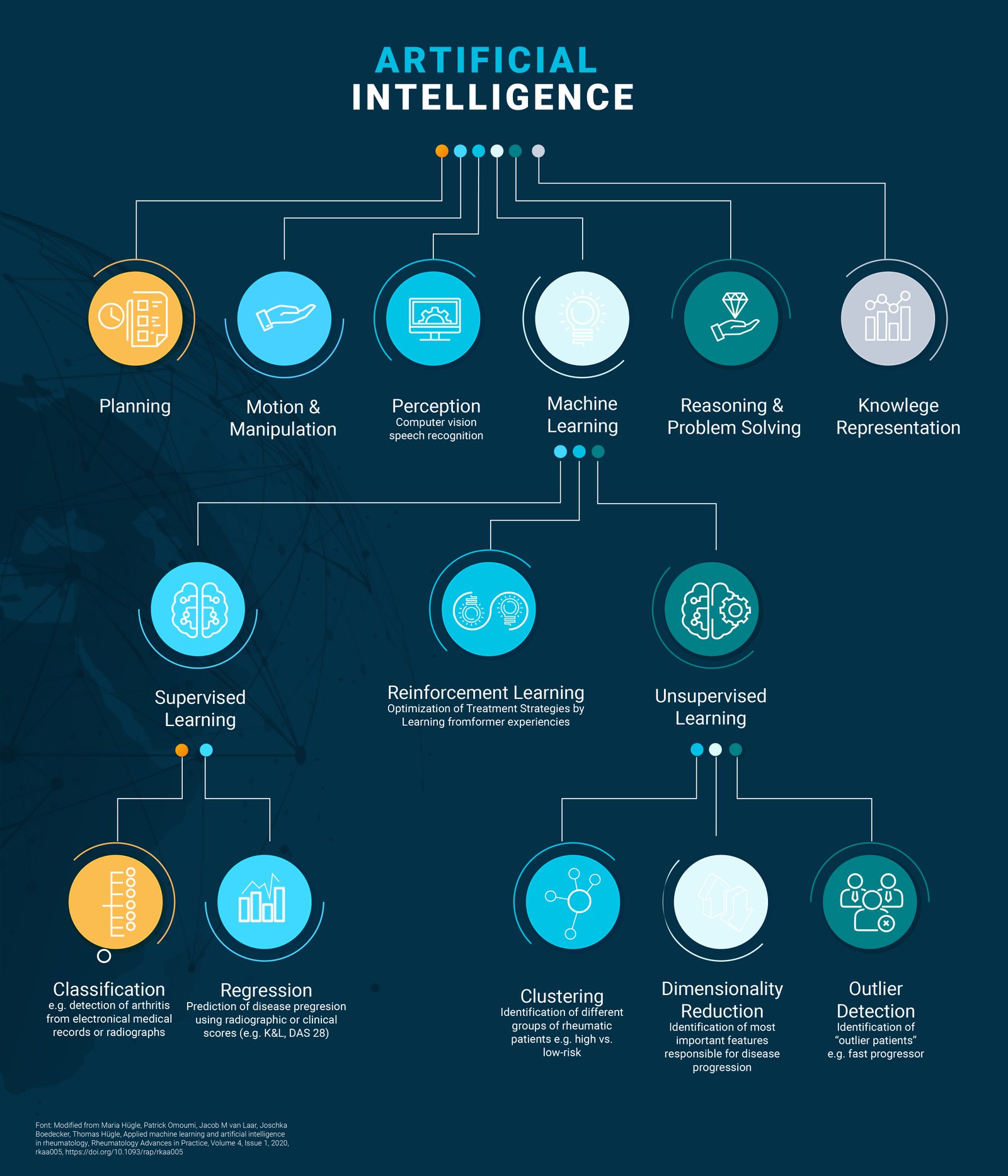
Source: Adapted from reference 6.
Some interesting and proven examples of AI use in rheumatology to date are:
- Algorithms for the identification of cases in medical records
A study of the development and validation of prediction algorithms demonstrated that machine learning methods are able to extract the records of patients with rheumatoid arthritis from electronic health record data with high precision, allowing research on very large populations for limited costs. The authors of the study highlight that their approach is language and center independent and could be applied to any type of diagnosis also considering the low cost from data already available in electronic health record systems. (8)
- Image analysis
Radiographs of sacroiliac joints are commonly used for the diagnosis and classification of axial spondyloarthritis. A study aimed at developing and validating an artificial neural network for the detection of definite radiographic sacroiliitis as a manifestation of axial spondyloarthritis (axSpA). (9)
The results of this study showed that the neural network achieved an excellent performance in the detection of definite radiographic sacroiliitis with an AUC of 0.97 and 0.94 for the validation and test datasets, respectively. Sensitivity and specificity for the cut-off weighting both measurements equally were 88% and 95% for the validation and 92% and 81% for the test set. (9)
- Capillaroscopy
Deep learning has proven to be a useful tool for nailfold videocapillaroscopy, resulting in the objective and homogeneous analysis of images taken with multiple devices.
Given the lack of homogenization for the procedure, especially in the way and place the images are taken, the counting of the capillaries, and the measuring of their size, a study was carried out to provide a deep learning-based software to obtain objective and exhaustive data for the whole nailfold without increasing the time or effort needed to do the examination or requiring expensive equipment. (10)
The automated software to count nailfold capillaries was designed through an exploratory image dataset (2,713 images with 18,000 measurements of 3 different types). Subsequently, application rules have been created to detect the morphology of nailfold videocapillaroscopy images, through a training set of images. The software reliability was evaluated with standard metrics used in the machine learning field for object detection tasks, comparing automatic and manual counting on the same NVC images. (10)
The results of the study showed that a mean average precision (mAP) of 0.473 was achieved for detecting and classifying capillaries and hemorrhages by their shape, and a mAP of 0.515 was achieved for detecting and classifying capillaries by their size. A precision of 83.84% and a recall of 92.44% in the identification of capillaries was estimated. (10)
- Disease classification and diagnosis
Psoriatic arthritis
Neural networks can be successfully trained to distinguish between seropositive rheumatoid arthritis (RA), seronegative RA and psoriatic arthritis (PsA) based on inflammatory patterns from hand MRI and also to test how psoriasis patients with subclinical inflammation fit into such patterns. (11)
MRI scans from 649 patients (135 seronegative RA, 190 seropositive RA, 177 PsA, 147 psoriasis) were fed into ResNet neural networks. The area-under-the-receiver-operating-characteristic curve (AUROC) was 75% for seropositive RA vs. PsA, 74% for seronegative RA vs. PsA and 67% for seropositive vs. seronegative RA. All MRI sequences were relevant for classification, however, when deleting contrast agent-based sequences the loss of performance was only marginal. The addition of demographic and clinical data to the networks did not provide significant improvements for classification. Psoriasis patients were mostly assigned to PsA by the neural networks, suggesting that a PsA-like MRI pattern may be present early in the course of psoriatic disease. (11)
Rheumatoid arthritis
A machine learning-based model that could predict changes in disease activity and predict the treatment response of patients with rheumatoid arthritis was developed as part of an open collaborative competition. Also, the analysis of patterns of joint involvement and an approach based in this type of artificial intelligence, allowed to develop a model that could predict the course of the disease in patients with juvenile idiopathic arthritis. (12)
Additionally, synovial transcriptomic analysis and a machine learning-based approach identified subgroups of patients with rheumatoid arthritis and allowed the development of a model that could predict the treatment response to TNF inhibitors. (12)
- Response to treatment
Artificial intelligence approaches for predicting response to treatment are under investigation in rheumatology, and many potential biological predictors of response to anti-TNFs have been identified. However, these studies are still at an exploratory stage, and the results will need validation before they can be implemented in clinical practice. (13)
A “promising” model has been described, to guide treatment decisions in clinical practice that is mainly based on clinical profiles with additional genetic information, taking into account that accurate prediction of treatment responses in patients with rheumatoid arthritis can provide valuable information on effective selection of medication.
Anti-TNF (Tumor Necrosis Factor) agents constitute an important second-line treatment after methotrexate, the classic first-line treatment for RA. However, patient heterogeneity prevents the identification of predictive biomarkers and to accurately model the responses to anti-TNF agents. This model described by the investigators proved to be useful for guiding treatment decisions in clinical practice as it is mainly based on clinical profiles with additional genetic information. (14)
Table 2
|
Project |
AI |
Application |
|
Identification of cases |
Machine learning |
Extract the records of patients with RA from electronic health record data |
|
Analysis of images |
Deep learning |
Detection of definite radiographic sacroiliitis as a manifestation of axial spondyloarthritis |
|
Disease classification |
Neural networks |
Distinguish between seropositive rheumatoid arthritis (RA), seronegative RA, and psoriatic arthritis (PsA) based on inflammatory patterns from hand MRI |
|
Response to treatment |
Big data |
Treatment decision guide based on clinical profiles with additional genetic information |
Source: Elaborated by the authors
ETHICS AND ARTIFICIAL INTELLIGENCE
Artificial intelligence and deep learning have, however, pragmatic and theoretical limitations that may complicate their rapid and widespread use in clinical practice. It is unlikely to replace rheumatologists or radiologists in image interpretation; instead, a hybrid solution that benefits from both, artificial and human intelligence will likely be developed.
Current applications of big data, data analytics and artificial intelligence in rheumatology, including registries, machine learning algorithms and consumer-facing platforms raise issues in four main bioethical areas: privacy and confidentiality, informed consent, impact on the medical profession and justice. Machine learning and big data aid diagnosis, treatment, and prognosis, but the final decision about the use of information from algorithms should be left to rheumatology specialists to maintain the promise of fiduciary obligations in the doctor-patient relationship. (15)
Regarding recent recommendations for the use of big data in rheumatology, EULAR sets out general principles addressing its use in rheumatic and musculoskeletal diseases. The points to consider cover aspects of data sources and data collection, privacy by design, data platforms, data sharing and data analyses, in particular through artificial intelligence and machine learning. (13)
The methodology for the formulation of these points was based on the results of the meeting of a multidisciplinary task force of 14 international experts with expertise from a range of disciplines including computer science and artificial intelligence. Based on a literature review of the current status of big data in rheumatic and musculoskeletal diseases and in other fields of medicine, the points were formulated. However, as the experts stated, these points may change as new evidence and advances in the different fields are presented. (13)
WHAT IS NEXT
In the future, machine learning will likely assist rheumatologists in predicting the course of the disease and identifying important disease factors. Even more interestingly, machine learning will likely be able to make treatment propositions and estimate their expected benefit (e.g., By reinforcement learning). Thus, in future shared decision-making will not only include the patient’s opinion and the rheumatologist’s empirical and evidence-based experience, but it will also be influenced by machine-learned evidence. (6)
Future applications of artificial intelligence in rheumatology could include examining associations between genotype and phenotype, as well as using AI to extract and analyze clinical data from electronic health records (EHR). EHRs contain large amounts of real-world patient data in both a structure form (information such as International Classification of Disease codes), as well as in a free-text arrangement (e.g., the narrative from the healthcare provider notes). (2)
Patient-reported outcomes, along with laboratory values, genetic and transcriptomic information (e.g., from synovial biopsies) and radiological data will increase the quality of machine learning evidence. Once machine learning learns from its own decisions, we really can speak of AI-supported medicine. To do so, an architecture of data collection, storage, processing, algorithms and, finally, integration in the clinical system and validation or AI support is necessary. (13)
In the span of their professional lives, a radiologist will read over 10 million images, a dermatologist will analyze 200,000 skin lesions, and a pathologist will review nearly 100,000 specimens. Now imagine a computer doing this work over days, rather than decades, and learning from and refining its diagnostic acumen with each new image. This is the capability that artificial intelligence will bring to medical care: the potential to interpret clinical data more accurately and mora rapidly than medical specialists. (16). It is only important to think that rheumatologists are also faced daily with a significant amount of images with which they must make decisions.
It is also noted that some of the challenges presented by big data and artificial intelligence include data sources and data collection: how to collect and store the data, ensuring ethics and privacy; how to interpret complex analytics data models; and what the clinical implications are: how to move from big data to clinical decision making. (13)
CONCLUSIONS
Machine learning has already shown clinically useful applications in rheumatology. It has the potential to support doctors in clinical and experimental medicine and to foster personalized medicine. For patients, this technology offers the possibility of greater transparency and autonomy. Integrated databases have the greatest potential to provide sufficient relevant information. (13)
Despite the widespread use of smart apps in healthcare, there are some challenges for their adoption. Acceptance of technology, especially for the diagnosis in clinical settings, concerns related to scalability, data integration and interoperability, security, privacy, and ethics of aggregated digital data are just some of the examples of the challenges ahead. For example, the early adoption of AI methods in online social media analytics revealed some ethical challenges that can potentially undermine the privacy and autonomy of individuals and cause stigmatization. (17)
It is clear for many that artificial intelligence could play an important role in addressing global inequities in health care, health system and population. However, the challenges in developing and implementing artificial intelligence applications need to be thoroughly analyzed, prior to widespread adoption and measurable impact. (13)
Equity must be central for the implementation of artificial intelligence in all healthcare systems. Large datasets are essential for the development of these technologies, but they must be representative of the population to ensure that everyone can benefit.
Minority groups are underrepresented in most datasets used for the development of artificial intelligence algorithms and the health challenges for these communities are less obvious to data science teams, which tend not to be representative of these populations. The rhetoric around artificial intelligence involves greater emphasis on personalized recommendations and individual action, however, this should not undermine the importance of continued collective action to address social and structural determinants of health. (14)
Without losing sight of its enormous potential, we could state that artificial intelligence is in its infancy in rheumatology. Further studies are required to refine and validate the approaches of AI before it can be used to guide rheumatic diseases management in clinical practice, but it is clear that machine learning is a young and emerging field in rheumatology.
Automated image recognition and lesion scoring on radiographs are likely to be among the first AI-assisted applications to enter routine clinical use.
ACKNOWLEDGMENTS
The authors of this paper for Global Rheumatology would like to thank Dr. Juan José Scali for his review prior to publication. Dr. Scali is a rheumatologist and osteologist, a master of Argentine Rheumatology.
REFERENCES
- Solomon DH, Rudin RS. Digital health technologies: opportunities and challenges in rheumatology. Nat Rev Rheumatol [Internet]. 2020 [citado el 12 de marzo de 2022];16(9):525–35. Disponible en: https://www.nature.com/articles/s41584-020-0461-x
- Fajardo E, Graf C. Inteligencia artificial, ¿transformación de la reumatología? - Parte I. Global Rheumatology [Internet]. Pan American League of Associations of Rheumatology (PANLAR); 2022 Apr 21; Available from: http://dx.doi.org/10.46856/grp.21.e115
- Artificial Intelligence in rheumatic diseases: Can it solve the treatment management puzzle? [Internet]. European Medical Journal. 2021. Disponible en: https://www.emjreviews.com/rheumatology/symposium/artificial-intelligence-in-rheumatic-diseases-can-it-solve-the-treatment-management-puzzle-j170121/
- Kothari S, Gionfrida L, Bharath AA, Abraham S. Artificial Intelligence (AI) and rheumatology: a potential partnership. Rheumatology (Oxford) [Internet]. 2019;58(11):1894–5. Disponible en: https://academic.oup.com/rheumatology/article/58/11/1894/5511607?login=tru
- Augmenting diagnostics visión with AI. Quer G, Muse ED, Nikzad N, Topol EJ, Steinhubl SR. Lancet 2017;3 90:22 1. https://doi.org/10.1016/S0140-6736(17)31764-6
- Hügle M, Omoumi P, Van Laar JM, Boedecker J, Hügle T. Applied machine learning and artificial intelligence in rheumatology, Rheumatology Advances in Practice, Volume 4, Issue 1, 2020, rkaa005, https://doi.org/10.1093/rap/rkaa005
- Stoel B. Use of artificial intelligence in imaging in rheumatology – current status and future perspectives. RMD Open 2020;6:e001063. doi:10.1136/rmdopen-2019-001063
- Maarseveen TD, Meinderink T, Reinders MJT, Knitza J, Huizinga TWJ, Kleyer A, Simon D, van den Akker EB, Knevel R. Machine Learning Electronic Health Record Identification of Patients with Rheumatoid Arthritis: Algorithm Pipeline Development and Validation Study. JMIR Med Inform. 2020 Nov 30;8(11):e23930. DOI: 10.2196/23930. PMID: 33252349; PMCID: PMC7735897.
- Bressem KK, Vahldiek JL, Adams L, et al. Deep learning for detection of radiographic sacroiliitis: achieving expert-level performance. Arthritis Research & Therapy. 2021 Apr;23(1):106. DOI: 10.1186/s13075-021-02484-0. PMID: 33832519; PMCID: PMC8028815.
- Gracia Tello B, Ramos Ibáñez E, Fanlo Mateo P, Sáez Cómet L, Martínez Robles E, Ríos Blanco JJ, Marí Alfonso B, Espinosa Garriga G, Todolí Parra J, Ortego Centeno N, Callejas Rubio JL, Freire Dapena M, Marín Ballvé A, Selva-O'Callaghan A, Guillén Del Castillo A, Simeón Aznar CP, Fonollosa Pla V. The challenge of comprehensive nailfold videocapillaroscopy practice: a further contribution. Clin Exp Rheumatol. 2021 Dec 16. DOI: 10.55563/clinexprheumatol/6usce8. Epub ahead of print. PMID: 34936544.
- Folle L, Bayat S, Kleyer A, Fagni F, Kapsner LA, Schlereth M, Meinderink T, Breininger K, Tascilar K, Krönke G, Uder M, Sticherling M, Bickelhaupt S, Schett G, Maier A, Roemer F, Simon D. Advanced neural networks for classification of MRI in psoriatic arthritis, seronegative, and seropositive rheumatoid arthritis, Rheumatology, 2022;, keac197, https://doi.org/10.1093/rheumatology/keac197
- Pandit, A., Radstake, T.R.D.J. Machine learning in rheumatology approaches the clinic. Nat Rev Rheumatol 16, 69–70 (2020). https://doi.org/10.1038/s41584-019-0361-0….
- Gossec L, Kedra J, Servy H, et al. EULAR points to consider for the use of big data in rheumatic and musculoskeletal diseases. Annals of the Rheumatic Diseases 2020;79:69-76
- Manrique de Lara A, Peláez-Ballestas I. Big data, and data processing in rheumatology: bioethical perspectives. Clin Rheumatol [Internet]. 2020;39(4):1007–14. Disponible en: http://dx.doi.org/10.1007/s10067-020-04969-w
- Guan Y, Zhang H, Quang D, Wang Z, Parker SCJ, Pappas DA, Kremer JM, Zhu F. Machine Learning to Predict Anti-Tumor Necrosis Factor Drug Responses of Rheumatoid Arthritis Patients by Integrating Clinical and Genetic Markers. Arthritis Rheumatol. 2019 Dec;71(12):1987-1996. DOI: 10.1002/art.41056. Epub 2019 Nov 4. PMID: 31342661.
- Mamiya, H., Shaban-Nejad, A. & Buckeridge, D. L. Online public health intelligence: ethical considerations at the big data era (eds. Shaban-Nejad, A., Brownstein, J. & Buckeridge, D. L.) Public Health Intelligence and the Internet. Lecture Notes in Social Networks 129–148 (Springer, Cham. 2017).
- Panch T, Pearson-Stuttard J, Greaves F, Atun R. Artificial intelligence: opportunities and risks for public health. Lancet Digit Health [Internet]. 2019;1(1):e13–4. Disponible en: https://www.thelancet.com/journals/landig/article/PIIS2589-7500(19)30002-0/fulltext